Carbon Nanotube Sensors Can Monitor Molecules In Your Body For Over a Year
This article is more than 2 years old
 MIT chemical engineering professor Michael Strano has devoted a lot of lab time, resources, and postdocs to figuring out ways to measure and better understand the role of nitric oxide in the body. Nitric oxide is a signaling molecule that exists in living cells, and while scientists know that it transfers information within the brain and helps coordinate bodily systems and functions such as the immune system, they’ve never really understood how nitric oxide works. They do know, though, that in cancerous cells, the nitric oxides levels are off, indicating that it has some role in the progression of cancer. In an attempt to learn more about that role, Strano and his lab have devised a measurement tool.
MIT chemical engineering professor Michael Strano has devoted a lot of lab time, resources, and postdocs to figuring out ways to measure and better understand the role of nitric oxide in the body. Nitric oxide is a signaling molecule that exists in living cells, and while scientists know that it transfers information within the brain and helps coordinate bodily systems and functions such as the immune system, they’ve never really understood how nitric oxide works. They do know, though, that in cancerous cells, the nitric oxides levels are off, indicating that it has some role in the progression of cancer. In an attempt to learn more about that role, Strano and his lab have devised a measurement tool.
The tool is a nitric oxide-monitoring sensor that can remain under the skin of a living being for over a year. The sensor would specifically monitor inflammation, which produces nitric oxide. The sensor is made of carbon nanotubes, which are about one nanometer (one billionth of a meter — the average germ is about 1,000 nanometers) thick. A carbon nanotube sensor works when scientists wrap them with a molecule that attaches to a specific target. When the binding occurs, the natural fluorescence of the carbon tube gets brighter or dimmer.
Such sensors can detect hydrogen peroxide or nerve gas, and Strano’s lab is currently working on one that can detect glucose or insulin levels in diabetics, making blood draws obsolete. For this experiment, the researchers wrapped the carbon nanotubes with a specific sequence of DNA. To inject the nanotube, the scientists embed it in a polymer gel and implant it — in their studies, it functioned under the skin of mice for 400 days, but it could last even longer. Once the sensors were in the body, the scientists shined infrared lasers on them, provoking a signal from the carbon nanotube. They believe that this kind of device could monitor cancer and other diseases, as well as immune reactions in people with artificial body parts or devices.
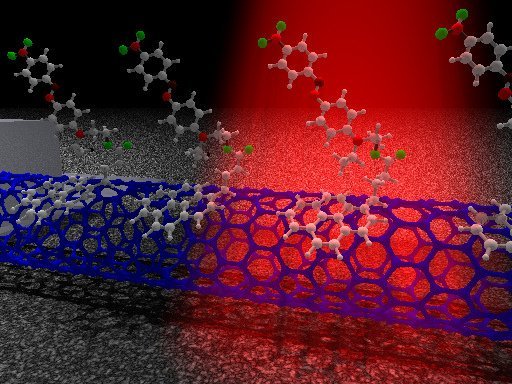
They also devised a short-term injectable version of the sensor that travels safely through the bloodstream (or at least it did in the mice they tested), and then the particles gather in the liver where they can then monitor nitric oxide. Scientists think that the injectable nanoparticle could reside in different organs for monitoring.
While nanosensors aren’t new, this is the first time scientists have been able to demonstrate that they can work in the body for a long period of time. Nanotechnology has long been at the forefront of medicine, shifting the paradigm from external monitoring of bodily functions and processing to internal monitoring. The question now is whether these sensors will survive the clinical trial phase and gain FDA approval. Jack Andraka, a 15-year-old TED speaker, devised a pancreatic cancer test using carbon nanotubes, which in itself makes a pretty convincing case for the possibilities. And a pretty convincing case for kid scientists.












